- Blog
- Nina tekken 3
- Old super mario bros fangame
- Farming simulator 19- mahindra retriever dlc
- The untethered soul pdf download
- 16 personalities premium profile download free estp
- Image capture not showing iphone photos
- Iso 13485 compliance
- Logitech web camera software download
- Driver zyrex m1115 win 7
- Define sap business one
Up your business, talk to our kwalitycert masters who are available for you to coach and on how to get your business and process certified with ISO 9001, ISO 14001, ISO 45001, ISO 22000,ISO 27001,ISO 20000-1 and HACCP. If you have one to know it should be this one. Kwalitycert is a catalyst for business and process excellence, your business and process excellence is guaranteed through ISO certification in Oman with kwlaitycert ISO 13485 is THE Standard for Medical Device Companies. It also helps you achieve compliance with the calibration requirements set out in ISO 13485:2016 Clause 7.6 (Control of monitoring and measuring equipment). Compliance to ISO 13485 requirementsĪ well-designed calibration program as described above helps you maintain the accuracy of your instruments. To summarize, calibration quantifies and controls errors and uncertainties within measurement processes and brings them to an acceptable level. Establishing traceability of the measurement to a National / International Standard, which is a mandatory requirement for most standards.Building trust, confidence and reliability in measurements.Keeping measurements within specification limits.Ensuring the reliability and consistency of the instrument.Adjusting or repairing an instrument which is out of calibration.

Restoring the accuracy of the instrument.

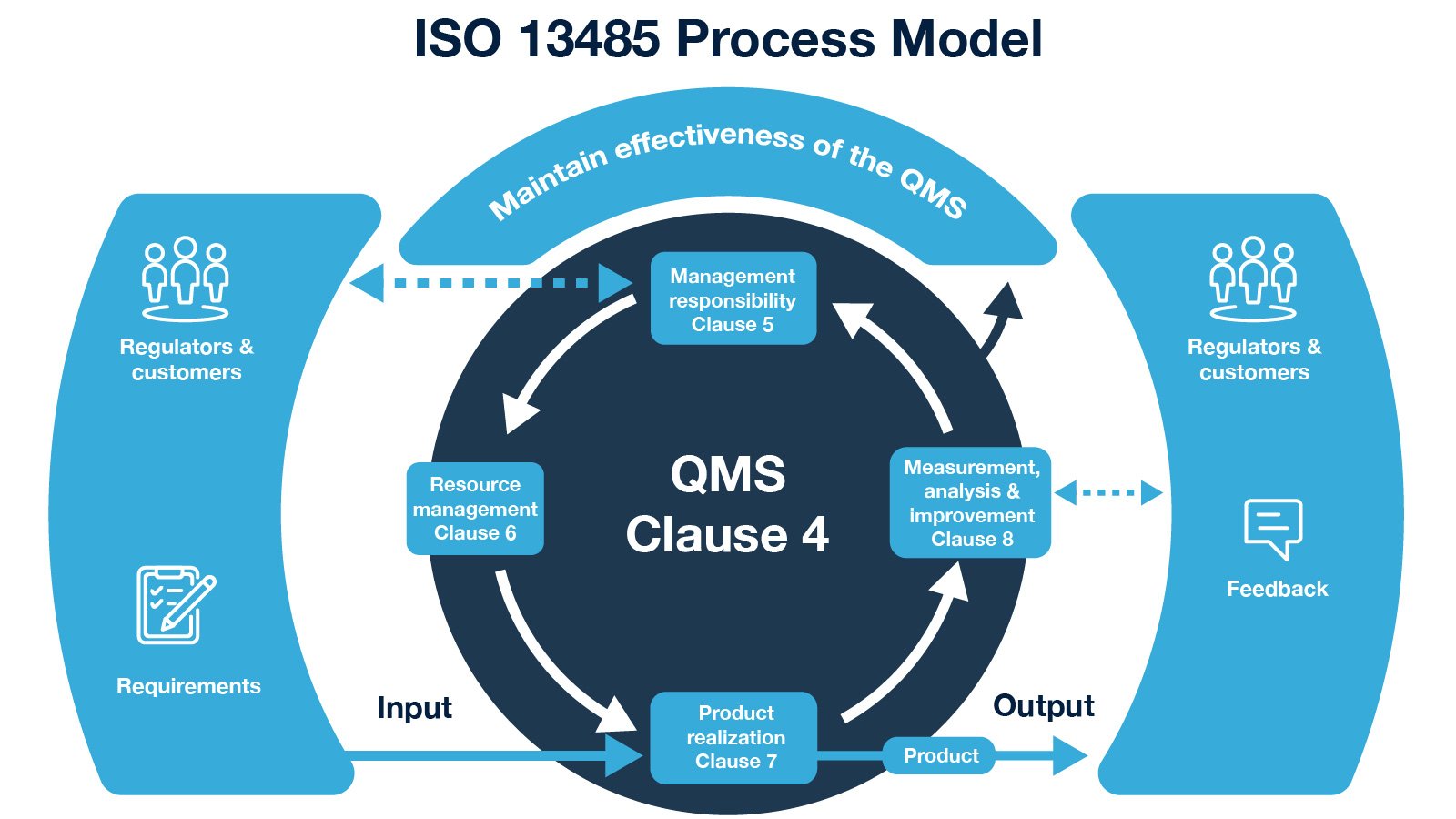
So, here’s what to expect in order to achieve compliance.Ĭlause 4 – Quality Management System (“Intersection”)Ĭlause 5 – Management Responsibility (“Highway”)Ĭlause 6 – Resource Management (“Roadway”)Ĭlause 7 – Product Realization (“Overpass”)Ĭlause 8 – Measurement, Analysis and Improvement (“Bridge”) What do you achieve by calibrating measuring and monitoring equipment? The remaining five clauses provide the framework for what is expected of organizations dedicated to medical device production.
ISO 13485 COMPLIANCE SOFTWARE
ISO 13485 Online Store: ISO 14971 Online Store: Support Technical Support: Support Policies: Software Registration: Software Installation: Licensing/Accounts. While ISO 13485 is based on the ISO 9001 process model concepts of Plan, Do, Check, Act, it is designed for regulatory compliance. Validation and 21 CFR Part 11 Compliance Certificate (ISO 13485 Software) IMSXpress Document Control and QMS Management Software. The standard includes eight clauses, three of which provide guidance on the intended use of the standard. ISO 13485 certifications adapt the ISO 9000 process-based model for a regulated medical device manufacturing environment. The ISO 13485 framework also forms the basis for auditing these same organizations, for both internal and external audits. Are you aiming to understand the ins and outs of ISO 13485? Are you the individual dedicated to implementation of this standard within your organization? Well, without reading the standard line by line, allow us to provide you with a “roadmap” of ISO 13485 certification in South Africa requirements and structure. Basically, ISO 13485 is like a quality management system for organizations involved in design, production, installation, and servicing of medical devices, with some other important requirements for good measure.
- Blog
- Nina tekken 3
- Old super mario bros fangame
- Farming simulator 19- mahindra retriever dlc
- The untethered soul pdf download
- 16 personalities premium profile download free estp
- Image capture not showing iphone photos
- Iso 13485 compliance
- Logitech web camera software download
- Driver zyrex m1115 win 7
- Define sap business one
